INDICATION
YCANTH (cantharidin) topical solution, 0.7% is indicated for the topical treatment of molluscum contagiosum in adult and pediatric patients 2 years of age and older.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS: None.
WARNINGS AND PRECAUTIONS:
- YCANTH is for topical use only. YCANTH is not for oral, mucosal, or ophthalmic use. Life threatening or fatal toxicities can occur if YCANTH is administered orally. Avoid contact with the treatment area, including oral contact, after treatment. Ocular toxicity can occur if YCANTH comes in contact with eyes. If YCANTH gets in eyes, flush eyes with water for at least 15 minutes.
- Local Skin Reactions: Reactions at the application site may occur, including vesiculation, pruritus, pain, discoloration, and erythema. Avoid application near eyes and mucosal tissue, and to healthy skin. If YCANTH contacts any unintended surface, or healthy skin, immediately remove. If severe local skin reactions occur, remove prior to the recommended 24 hours after treatment.
- YCANTH is flammable, even after drying. Avoid fire, flame or smoking near lesion(s) during treatment and after application until removed.
ADVERSE REACTIONS:
The most common (incidence ≥1%) reactions are the following local skin reactions at the application site: vesiculation, pain, pruritus, scabbing, erythema, discoloration, application site dryness, edema, and erosion. Local skin reactions at the application site were observed in 97% of subjects treated with YCANTH during clinical trials. These local skin reactions are expected and related to the anticipated blistering response of the skin to cantharidin.
DRUG INTERACTIONS:
No studies evaluating the drug interaction potential of cantharidin have been conducted.
USE IN SPECIFIC POPULATIONS:
Pregnancy: There are no available data with use of YCANTH in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Given that systemic exposure to cantharidin following topical administration is low, maternal use is not expected to result in fetal exposure to the drug.
Lactation: Avoid application of YCANTH topical solution to areas with increased risk for potential ingestion by or ocular exposure to the breastfeeding child.
OVERDOSAGE:
Oral ingestion of cantharidin has resulted in renal failure, blistering and severe damage to the gastrointestinal tract, coagulopathy, seizures, and flaccid paralysis.
Please see full Prescribing Information.
To report SUSPECTED ADVERSE REACTIONS, contact Verrica Pharmaceuticals Inc. at 1-877-VERRICA (1-877-837-7422), or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. Local skin reactions are expected and should be reported if they are severe.
References:
1. Eichenfield LF, Siegfried E, Kwong P, et al. Pooled results of two randomized phase III trials evaluating VP-102, a drug-device combination product containing cantharidin 0.7% (w/v) for the treatment of molluscum contagiosum. Am J Clin Dermatol. 2021;22(2):257-265. doi:10.1007/s40257-020-00570-8
2. YCANTH (cantharidin) topical solution 0.7% Prescribing Information, Verrica Pharmaceuticals Inc., 2024.
3. Data on file. Verrica Pharmaceuticals Inc., 2024.
4. Eichenfield LF, McFalda W, Brabec B, et al. Safety and efficacy of VP-102, a proprietary, drug-device combination product containing cantharidin, 0.7% (w/v), in children and adults with molluscum contagiosum: two phase 3 randomized clinical trials. JAMA Dermatol. 2020;156(12):1315-1323. doi:10.1001/jamadermatol.2020.3238
5. Olsen JR, Gallacher J, Finlay AY, Piguet V, Francis NA. Time to resolution and effect on quality of life of molluscum contagiosum in children in the UK: a prospective community cohort study. Lancet Infect Dis. 2015;15(2):190-195. doi:10.1016/S1473-3099(14)71053-9
6. Centers for Disease Control and Prevention. Molluscum contagiosum: transmission. Accessed March 12, 2024. https://www.cdc.gov/poxvirus/molluscum-contagiosum/transmission.html
7. American Academy of Pediatrics. Molluscum contagiosum. In: Kimberlin DW, Barnett ED, Lynfield R, Sawyer MH, eds. Red Book: 2021-2024 Report of the Committee on Infectious Diseases. American Academy of Pediatrics; 2021:535-537.
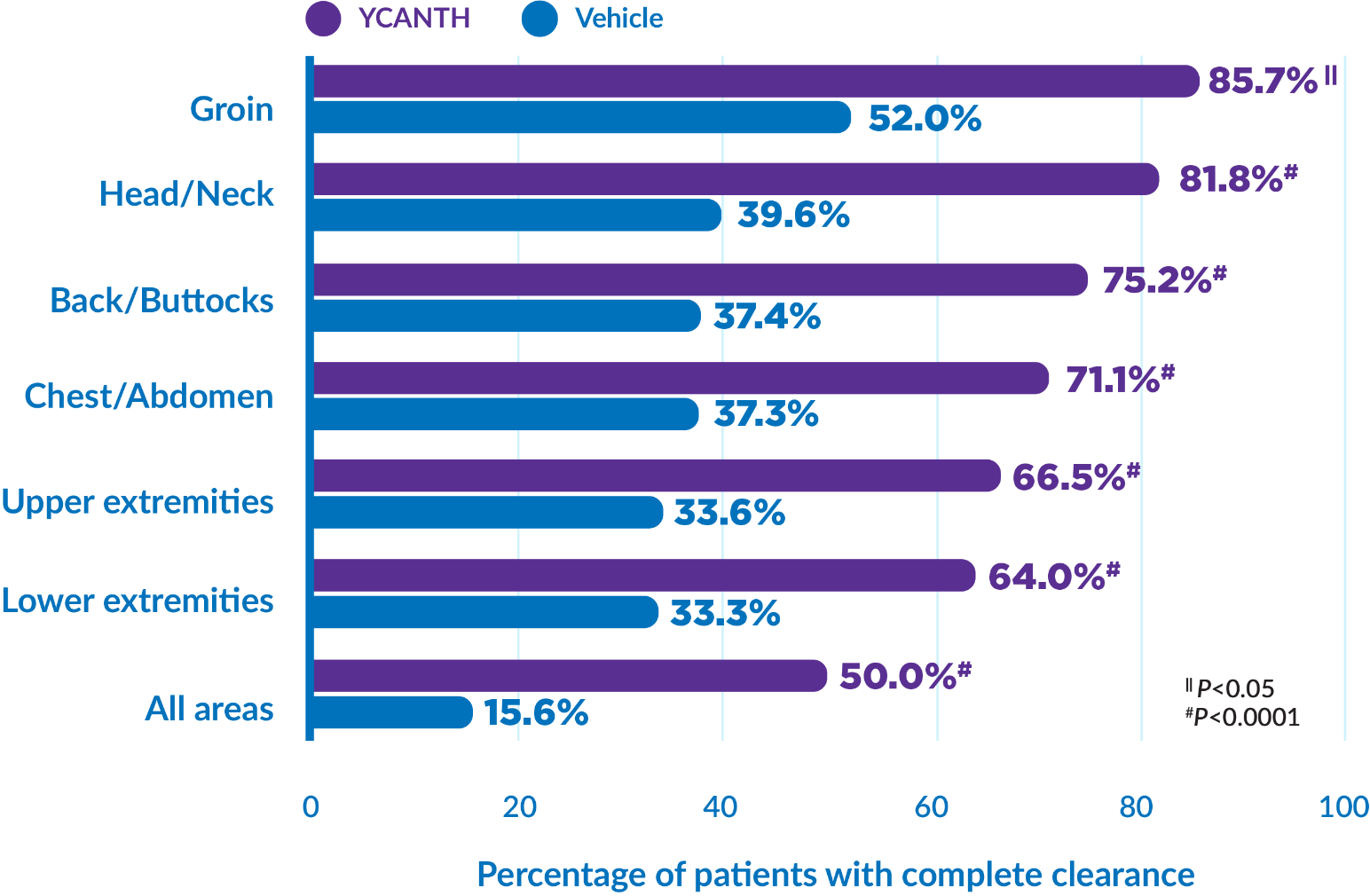
8. Eichenfield LF, Kwong P, Gonzalez ME, et al. Safety and efficacy of VP-102 (cantharidin, 0.7% w/v) in molluscum contagiosum by body region: post hoc pooled analyses from two phase III randomized trials. J Clin Aesthet Dermatol. 2021;14(10):42-47.
CAMP‑1/CAMP‑2=Cantharidin Application in Molluscum Patients-1 and -2; EOS=end of study; ITT=intent to treat; SD=standard deviation.